Slater argued that there must be a connection between empirically determined atomic and ionic size with the wave functions of atoms and ions and pointed out a good correlation between the atomic radii and the distance of maximum charge density in the outermost electron shell of the atom. The fact that each electron in a many electron system should have one-electron function is a pre-quantum mechanical thinking when it was pointed out that many spectroscopic facts could be rationalized if energy levels were identified with those of a single electron moving in a central field. The inherent approximation of the SCF method suggests that each electron have its own one-electron function or orbital in many–electron situation. Also, there is no means of verifying the merit of a set of radii determined experimentally or calculated theoretically because, there is no experimental value of the sizes of atoms and ions. But such radii data though useful in reproducing the inter atomic/ionic distances for the particular purposes but not useful in computing the radial dependent absolute properties of atoms. But invariably the text books display one or the other chart of atomic and ionic radii and recently there has been a proliferation of the tables and data of atomic and ionic radii with bewildering array of terms including bonded, non-bonded, ionic, covalent and van der Waals radii. 
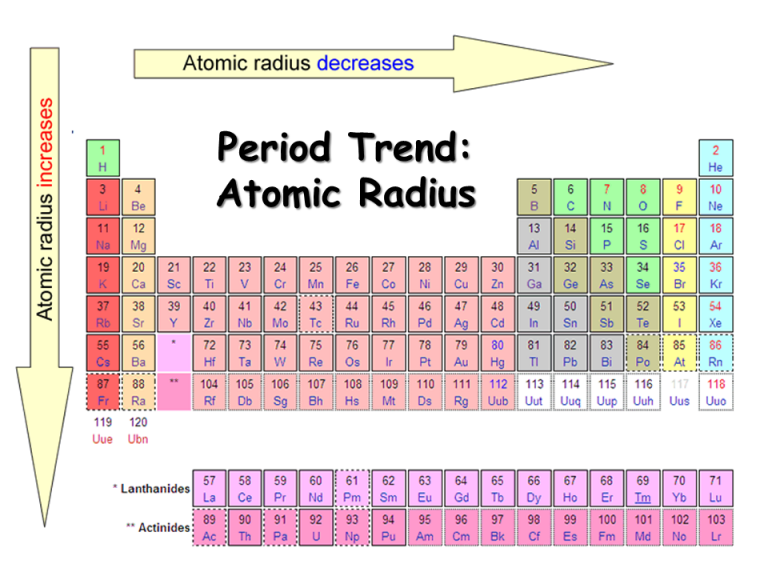
The text books dealing with this size aspects of atoms or ions never make the position distinctly clear as to the meaning and purport of the terms ‘atomic’ or ‘ionic’ radii and the term ‘radii’ is kept vague. Unfortunately this very important property –– the size concept of atoms and ions is heavily underplayed. However, the inter atomic or inter ionic distance depend upon the crystal type, its allotropic modification, co-ordination number, temperature etc. Approximate additivity of atomic and ionic radii were noted by early investigators like Goldschmidt et al, Pauling, Zachariasen, and Bragg. The crystal chemists have tried for many years to treat atoms and ions as hard spheres and published series of atomic and ionic radii with the only significance that such atomic and ionic radii, when added, reproduce the minimum distance of separation between atoms and ions respectively in crystal lattices. The atomic and ionic radii values are important input in many calculations of size dependent physico–chemical properties of isolated atoms. The atomic size has been also an important periodic property of the elements. The atomic volume curve of Lother Meyer was a striking example of periodicity of physical properties of elements. The periodicity of chemical and physical properties of the elements has been recognized from the date of early history of chemistry. The concept of atomic and ionic radii has been found to be very useful in understanding, explaining, correlating and even predicting many physico-chemical properties of atoms, ions and molecules. A simple method of computing the absolute size of atoms has been explored and a large body of known material has been brought together to reveal how many different properties correlate with atomic size. 
The calculated global hardness and atomic polarizability of a number of atoms are found to be close to the available experimental values and the profiles of the physical properties computed in terms of the theoretical atomic radii exhibit their inherent periodicity. 
The radii are used to calculate a number of size dependent periodic physical properties of isolated atoms viz., the diamagnetic part of the atomic susceptibility, atomic polarizability and the chemical hardness. The computed sizes qualitatively correlate with the absolute size dependent properties like ionization potentials and electronegativity of elements. The d-block and f-block contractions are distinct in the calculated sizes. The set of theoretical radii are found to reproduce the periodic law and the Lother Meyer’s atomic volume curve and reproduce the expected vertical and horizontal trend of variation in atomic size in the periodic table. (Credit: Christopher Auyeung Source: CK-12 Foundation License: CC BY-NC 3.A set of theoretical atomic radii corresponding to the principal maximum in the radial distribution function, 4πr 2R 2 for the outermost orbital has been calculated for the ground state of 103 elements of the periodic table using Slater orbitals. \): Atomic radii of the representative elements measured in picometers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
